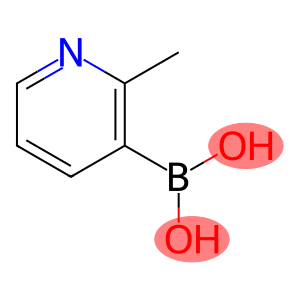
2-Methyl-3-Pyridinylboronic Acid
2-methylpyridine-3-boronic acid
CAS: 899436-71-6
Molecular Formula: C6H8BNO2
2-Methyl-3-Pyridinylboronic Acid - Names and Identifiers
| Name | 2-methylpyridine-3-boronic acid |
| Synonyms | 3-Borono-2-methylpyridine 2-PICOLINE-3-BORONIC ACID 2-Methyl-3-pyridineboronic acid 2-methylpyridine-3-boronic acid 2-Methylpyridin-3-ylboronic acid 2-METHYL-3-PYRIDINYLBORONIC ACID Boronic acid, B-(2-Methyl-3-pyridinyl)- |
| CAS | 899436-71-6 |
| InChI | InChI=1/C6H8BNO2/c1-5-6(7(9)10)3-2-4-8-5/h2-4,9-10H,1H3 |
2-Methyl-3-Pyridinylboronic Acid - Physico-chemical Properties
| Molecular Formula | C6H8BNO2 |
| Molar Mass | 136.94 |
| Density | 1.18±0.1 g/cm3(Predicted) |
| Boling Point | 303.6±44.0 °C(Predicted) |
| pKa | 4.08±0.58(Predicted) |
| Storage Condition | Inert atmosphere,Store in freezer, under -20°C |
| Refractive Index | 1.527 |
2-Methyl-3-Pyridinylboronic Acid - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R41 - Risk of serious damage to eyes R37/38 - Irritating to respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
| Hazard Class | IRRITANT |
2-Methyl-3-Pyridinylboronic Acid - Reference Information
| Application | 2-methylpyridine-3-boronic acid is an organic intermediate, it can be prepared from 3-bromo-2-methylpyridine and triisopropyl borate in one step. 2-methylpyridine-3-boronic acid can be used to participate in the Suzuki reaction. |
| preparation | to 3-bromo-2-methylpyridine (4.00g,23 mmol) at -78°C butyl lithium (17mL,28 mmol) was added dropwise to a solution of triisopropyl borate (6.40 mL,28 mmol) and 50 mL/1 toluene/THF(4/1,50 mL). The mixture was heated to -70 °c over 30 minutes and then to 20 °c. HCl(2m) was added to bring the solution to pH 1. Water (20ml) was then added and the mixture was extracted with toluene. The aqueous layer was neutralized with 1M NaOH and extracted with dichloromethane. The aqueous layer was concentrated to dryness and the white solid was washed with dichloromethane. The combined organic layers were dried over sodium sulfate, filtered and concentrated to give 2.10g of 2-methylpyridine-3-boronic acid as a yellow oil. ES = 138.2(M H). |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2-Methyl-3-Pyridinylboronic Acid Visit Supplier Webpage Request for quotationCAS: 899436-71-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-methylpyridine-3-boronic acid Request for quotation
CAS: 899436-71-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 899436-71-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-METHYLPYRIDINE-3-BORONIC ACID Request for quotation
CAS: 899436-71-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 899436-71-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-Methyl-3-Pyridinylboronic Acid Visit Supplier Webpage Request for quotation
CAS: 899436-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 899436-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Methyl-3-Pyridinylboronic Acid Visit Supplier Webpage Request for quotationCAS: 899436-71-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-methylpyridine-3-boronic acid Request for quotation
CAS: 899436-71-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 899436-71-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-METHYLPYRIDINE-3-BORONIC ACID Request for quotation
CAS: 899436-71-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 899436-71-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-Methyl-3-Pyridinylboronic Acid Visit Supplier Webpage Request for quotation
CAS: 899436-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 899436-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



